|
Addition of the solid causes an upward displacement of the liquid, resulting in a new volume reading. 
The solid is added to the cylinder, and the total mass is weighed again to determine the mass of the solid. A graduated cylinder containing a known volume of liquid (in which the solid is insoluble) is tared. 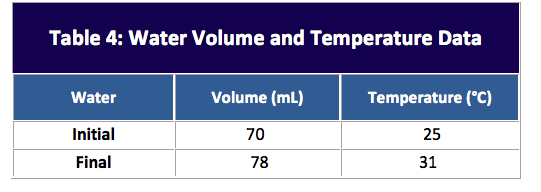
Instead of directly measuring dimensions or using glassware like a volumetric flask, it is necessary to make use of a liquid displacement method to measure the volume of an irregularly shaped solid. It is inaccurate, for example, to determine the volume of a powder by measuring its dimensions. Most solid substances are irregularly shaped, which complicates volume determination. The measured density is the ratio of the measured mass to the volume indicated on the flask. The container is filled to the line with the liquid sample and weighed again after the empty flask has been tared. The container to hold the sample (also used to measure volume) is weighed and tared, so only the sample mass appears on the balance display when the sample is added to the container.įor liquids, this container is typically a volumetric flask, which has one marking that corresponds to a specific volume. Mass is typically measured using an analytical balance, a precise instrument that relies on the force exerted by the sample due to gravity. To measure the density of a sample of a substance, it is necessary to measure its mass and volume. Thus, density can be used to identify an unknown pure substance if a list of reference densities is available, and the experimenter can choose a convenient amount of substance to work with when measuring density. At constant temperature and pressure, the density of a substance is constant.ĭensity is an intensive property of a substance that doesn’t depend on the amount of substance present. The density of a substance is the ratio of its mass to its volume. Using a graduated cylinder, analytical balance, and water as the displaced liquid, the density of zinc metal can be determined.īy definition, all matter has mass and occupies volume. Using a volumetric flask and an analytical balance, the density of ethanol can be determined. 
This demonstration illustrates the methods for measuring the density of solids and liquids. The volume of an irregularly-shaped solid can be measured by submersion in a liquid - the difference in volume caused by addition of the solid is equal to the volume of the solid. 
As liquids can flow and take the shapes of their containers, glassware such as a graduated cylinder or volumetric flask can be used to measure the volume of a liquid. For both solids and liquids, a balance can be used to measure mass however, methods for determining volume are different for solids and liquids. To measure the density of a sample of material, both the mass and volume of the sample must be determined. Because the density of a substance does not depend on the amount of substance present, density is an “intensive property”. Density is expressed in units of mass per volume, such as g/mL or kg/m 3. The ratio of the mass of a substance to its volume is known as the mass density or, simply, the density of the substance. Michael Evans - Georgia Institute of Technology
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
